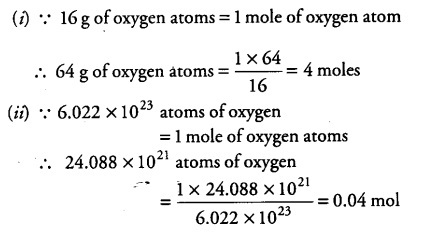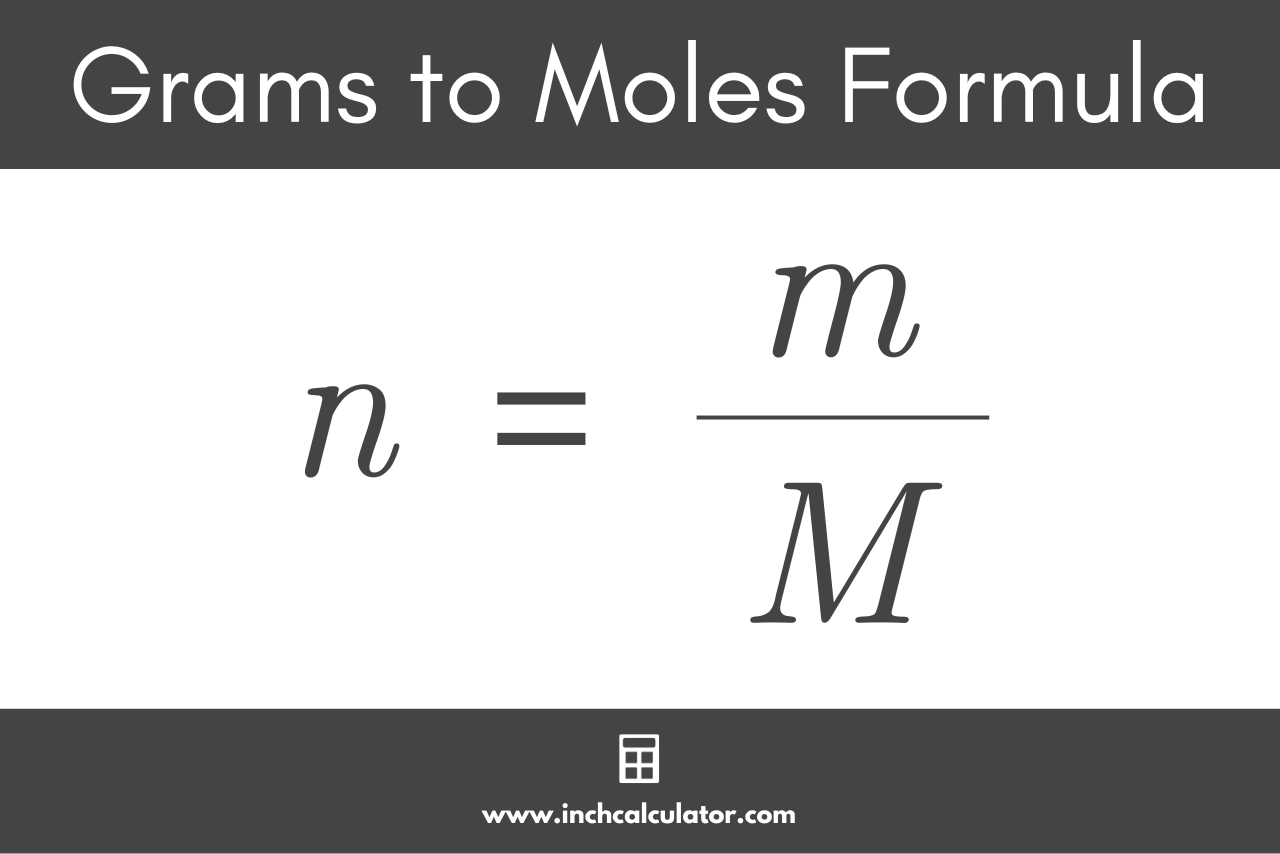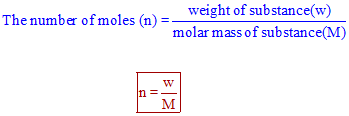How to Find Moles of Product from Moles of Reactant using a Chemical Equation | Chemistry | Study.com

Calculate the number of moles of gas present in the container of volume 10 L at 300 K. If the manometer containing glycerin shows 5 m difference in level as shown diagram.

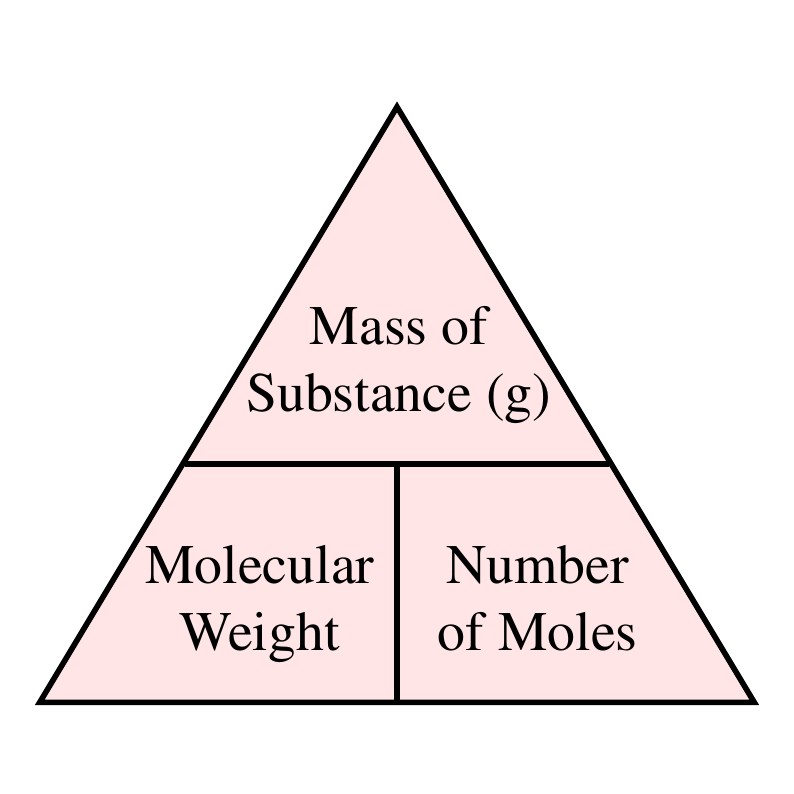
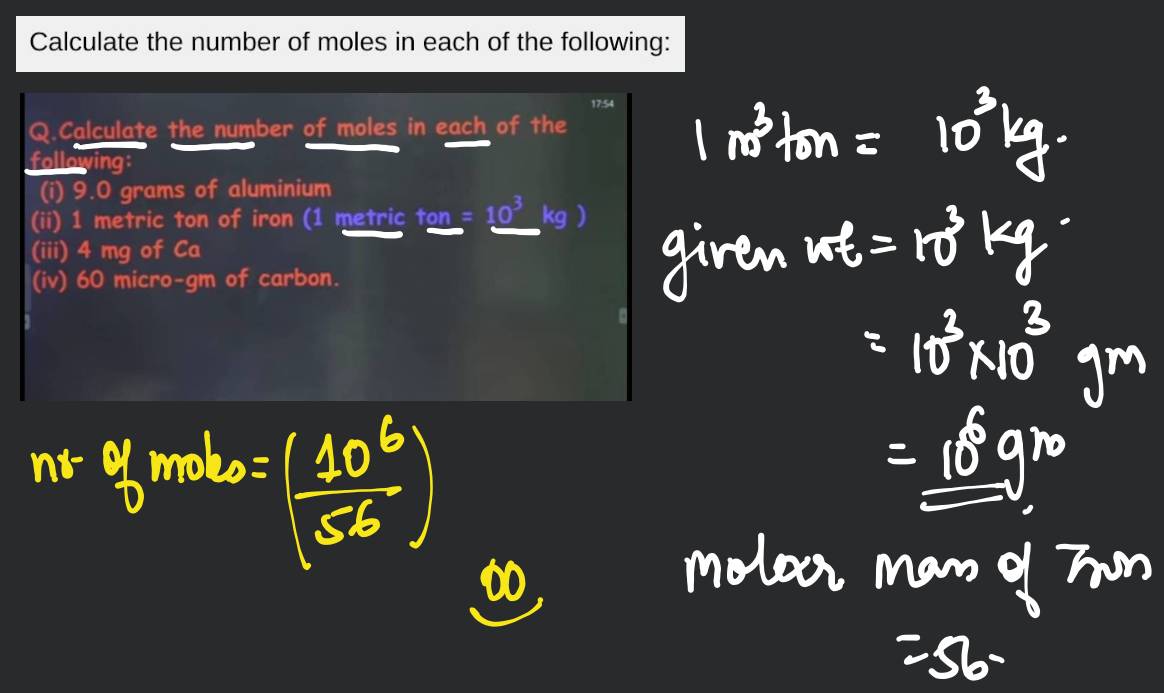
Calculate the number of moles the following:52 g of He (finding mole from mass)12.044times 10^ {23} number of He atoms (finding mole from number of particles)
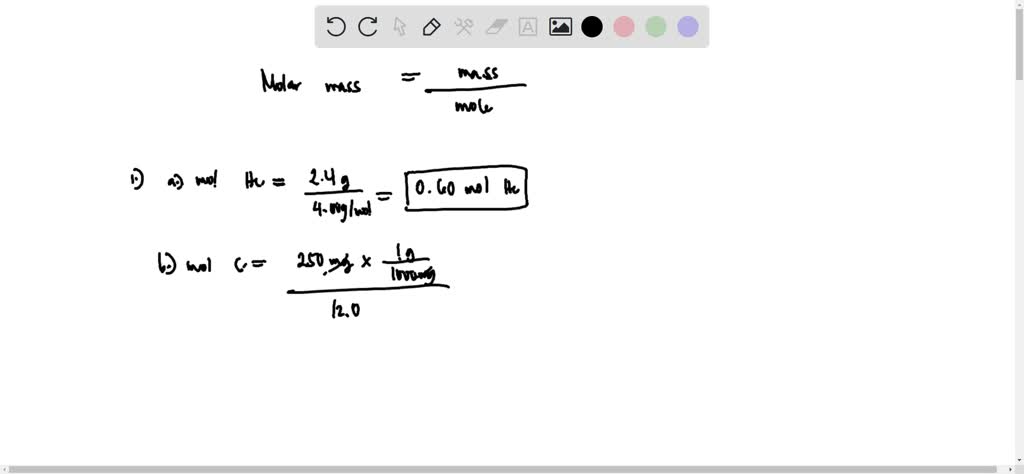
SOLVED: Q1. Calculate the number of moles of each substance in samples with the following masses: a) 2.4 g of He b) 250 mg of Carbon c) 15 g of sodium chloride

Mole Review 1.) Calculate the number of moles in 60.4L of O2. 2.) How many moles are there in 63.2g… | Chemistry education, Chemistry basics, How to calculate moles