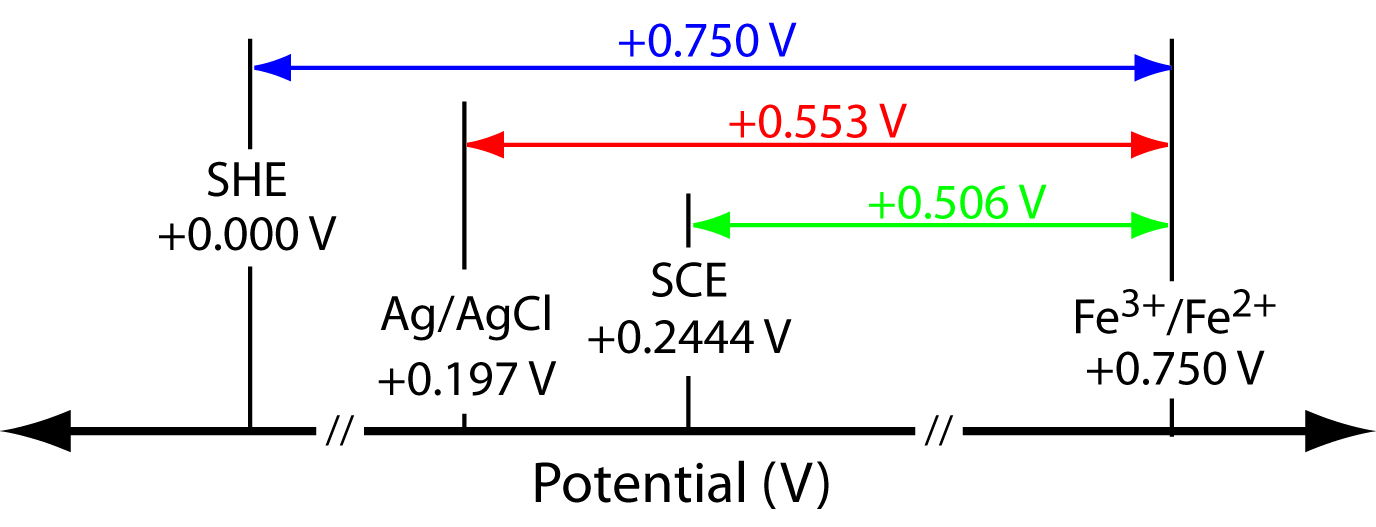
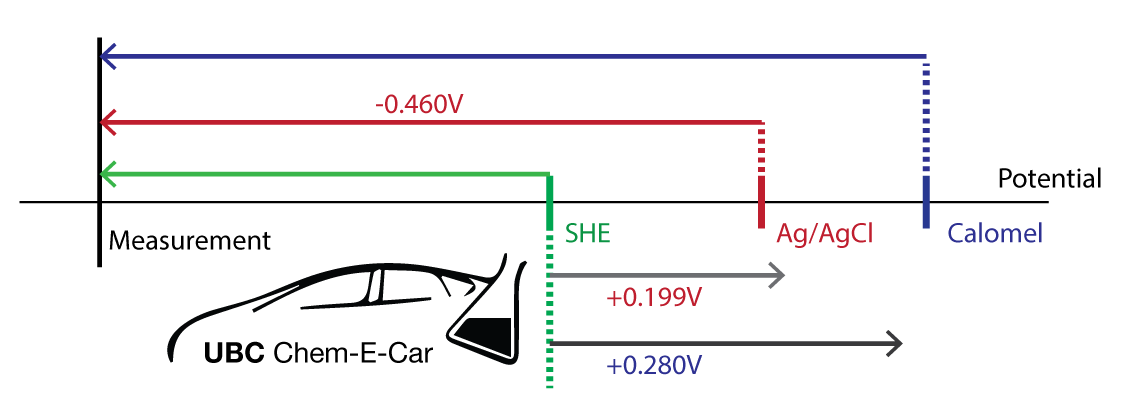
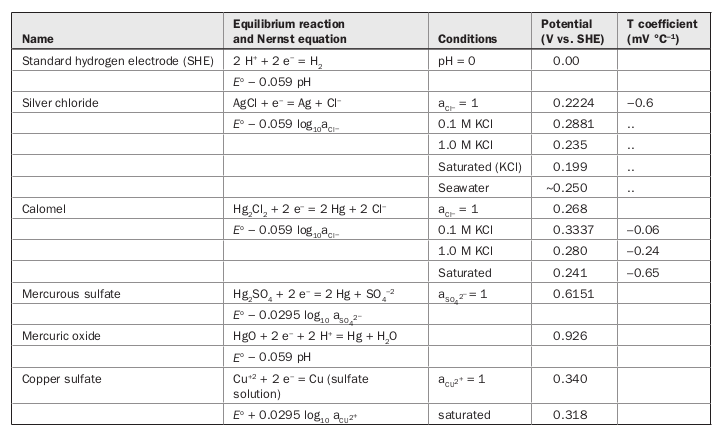
SOLVED: Unanswered Question 7 0 / 1 pts Consider the following redox reaction: MnO- + SFe ++8Ht Mn?+ + SFe ++4H,O A0.568 2-g sample of Fe(NH4)2(S0412 * 6H20 (FM 392.14) is dissolved
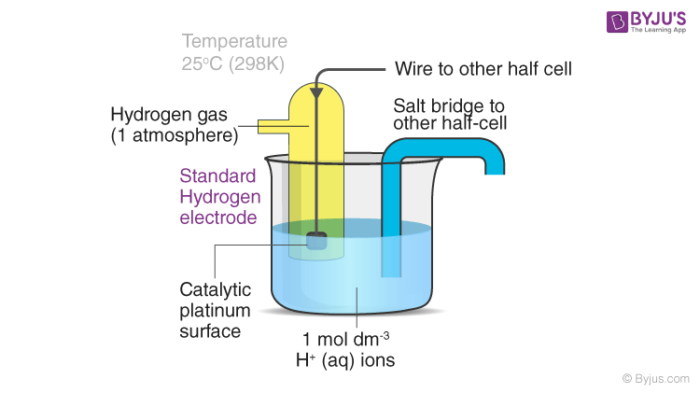
Standard Hydrogen Electrode - Definition, Construction, and Labelled Diagram of Standard Hydrogen Electrode

How can I convert RHE from the measured potentials in different reference electrodes and electrolytes? | ResearchGate