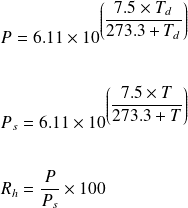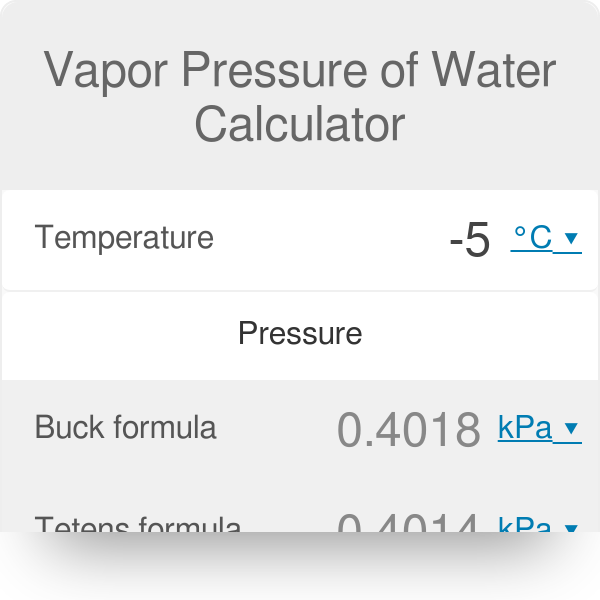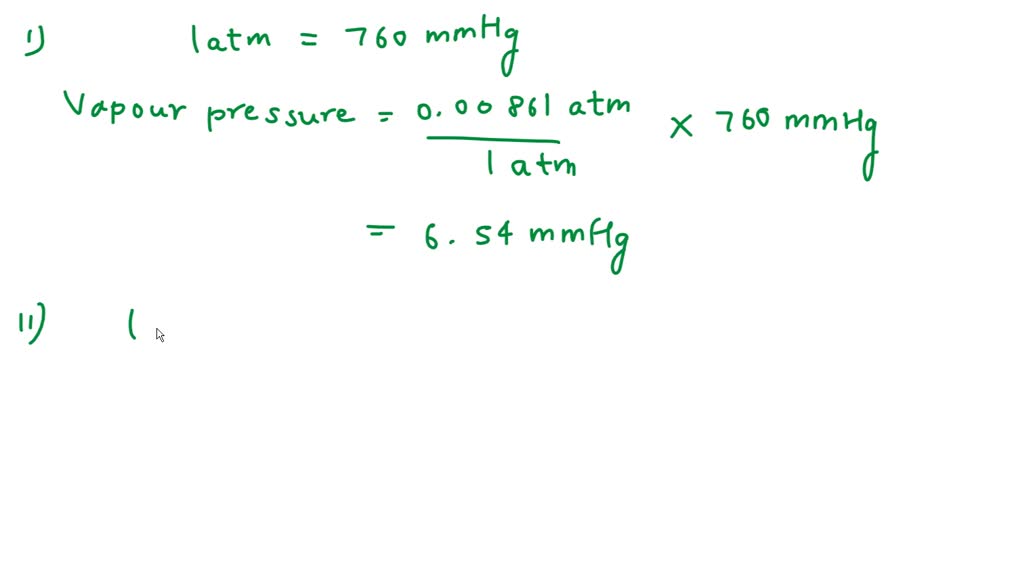
SOLVED: The vapor pressure of water at 5°C is 0.00861 atm. Calculate the vapor pressure in mmHg and torr. Round each of your answers to 3 significant digits. mmHg? torr?

SOLVED: Over a water surface, the saturation vapor pressure (es) is related to the air temperature (T) by the following equation: 17.27T es = 611exp(237.3 + T) where es is in pascals

a) Vapour pressure of benzene is 200 mm of Hg. When 2 gram of a non-volatile solute dissolved in 78 gram benzene. Benzene has vapour pressure of 195 mm of Hg. Calculate

A Simple Accurate Formula for Calculating Saturation Vapor Pressure of Water and Ice in: Journal of Applied Meteorology and Climatology Volume 57 Issue 6 (2018)
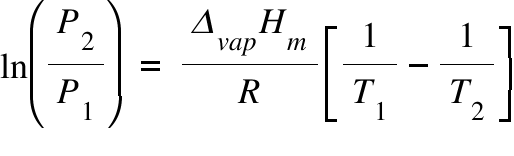
Correlation of Vapor Pressure at Different Temperatures by Clausius Clapeyron Equation Calculator | Calistry
Calculate vapour pressure of 0.1M urea soln. Vapour pressure of water at the given temperature is 20 torr. Assume molality and molality to be equal.